The presence of the delocalized electrons makes benzene particularly stable.īenzene resists addition reactions because those reactions would involve breaking the delocalization and losing that stability.īenzene is represented by this symbol, where the circle represents the delocalized electrons, and each corner of the hexagon has a carbon atom with a hydrogen attached. There are delocalized electrons above and below the plane of the ring. 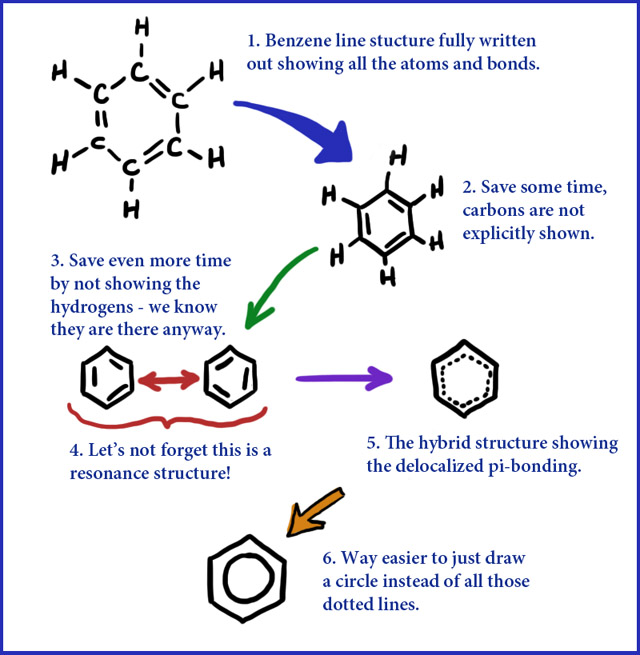
The carbons are arranged in a hexagon, and he suggested alternating double and single bonds between them. Kekul was the first to suggest a sensible structure for benzene. All of the carbon-carbon bonds have exactly the same lengths - somewhere between single and double bonds. 7: Delocalized Electrons and Their Effect on Stability, Reactivity, and pKa (More About Molecular Orbital Theory) 7.2: The Bonding in Benzene. The benzene molecule is best described by the MO theory.E. Both the bonding and the bonding are delocalized.D. The six carbon atoms form a perfectly regular hexagon. Science Chemistry Chemistry questions and answers The following statements concern molecules that require resonance. The other molecular orbitals are almost never drawn.īenzene, C 6H 6, is a planar molecule containing a ring of six carbon atoms, each with a hydrogen atom attached. Lots of compounds in nature contain 'benzene units' within larger structures. Benzene is the common example of an aromatic compound. Benzene, C 6 H 6, is often drawn as a ring of six carbon atoms, with alternating double bonds and single bonds: This simple picture has some complications, however. Aromatics are a special class of delocalized compounds. This diagram shows one of the molecular orbitals containing two of the delocalized electrons, which may be found anywhere within the two "doughnuts". Aromatic rings (also known as aromatic compounds or arenes) are hydrocarbons which contain benzene, or some other related ring structure. The delocalization of the p-orbital carbons on the sp 2 hybridized carbons is what gives the aromatic qualities of benzene. It will also go into detail about the unusually large resonance energy due to the six conjugated carbons of benzene. 
resonance in Lewis dot structures using the benzene Lewis dot structure. This section will try to clarify the theory of aromaticity and why aromaticity gives unique qualities that make these conjugated alkenes inert to compounds such as Br 2 and even hydrochloric acid. of bonding that describes the delocalization of electrons within molecules. \)Īmong the many distinctive features of benzene, its aromaticity is the major contributor to why it is so unreactive.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
